Physical Chemistry of Layered Nanomaterials - Group Prof. Dr. Claudia Backes

RESEARCH
Two dimensional nanomaterials beyond graphene such as transition metal dichalcogenides (TMDs) or black phosphorus (BP) have received considerable attention the past years because of their interesting physical properties and applications potential in a number of areas from (opto-)electronics to electrocatalysis to energy conversion. However, in order for these materials to be used, large quantities of controlled sizes need to be made available. Exfoliation in suitable liquids seems to be a promising way forward, in particular because hybrids and composites of different materials can be formed rather easily. In addition, it will be necessary to controllably modify surface and electronic properties for example by chemical functionalisation. If this was to be achieved, layered nanomaterials have the potential to become building blocks to form hierarchical, multifunctional architectures tailored to specific applications. Our research is dedicated to this goal starting from making dispersions with predefined sizes and thicknesses from a range of bulk crystals by developing novel size selection techniques to functionalising the nanomaterials and to form and analyse hybrid materials. To achieve this, we identify spectroscopic fingerprints and analytical protocols to track both nanosheet sizes and functionalisation.
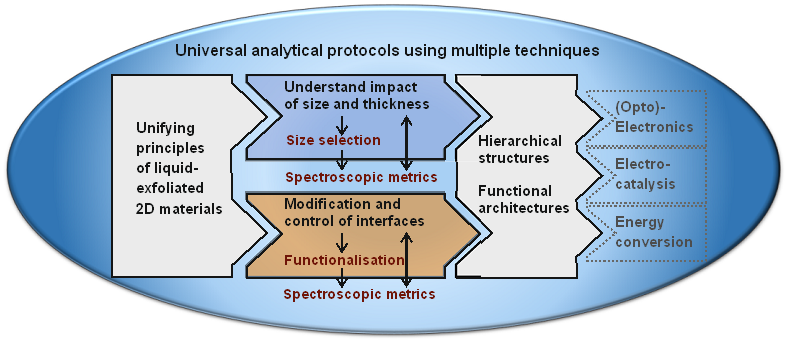
Materials and exfoliation

As starting materials, we use a broad range of layered bulk crystals of mainly transition metal dichalcogenides (e.g. MoS2, WS2, WSe2, MoTe2), but also III-VI metal chalcogenide semiconductors (e.g. GaS, InSe), metal oxides (e.g. MoO3) or black phosphorus. It has been shown that these materials can be delaminated or exfoliated to mono- and few-layered species in liquids by sonication or shear. Suitable solvents, surfactants or polymers can be used to stabilise the nanosheets from reaggregation in the dispersion. This gives access to a whole host of nanostructures that can all be processed from liquids which is hugely beneficial over other production techniques
Spectroscopic properties
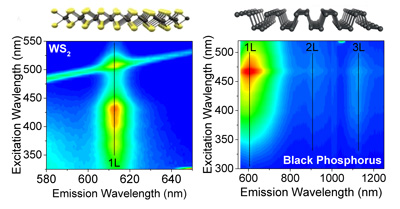
Size selection

A major disadvantage of liquid exfoliation is that nanosheets are polydisperse with respect to layer number or lateral dimension. This also means that monolayer contents are intrinsically very low. However, due to the processeability from liquids, a number of size selection techniques that are available for other materials (for example carbon allotropes or nanoparticles) can be adapted. We mainly focus on developing improved centrifugation-based methods, but also explore alternative routes by chromatography or size selective precipitation. The goal is not only to increase the monolayer contents, but rather to control both nanosheet length and thickness independently -over the broadest possible range while minimising material wastage.
Functionalisation
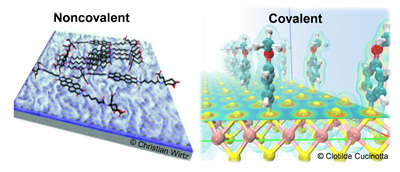
Up until now, the chemistry of the liquid-exfoliated 2D materials is only little explored. In particular, wet chemical functionalization sequences to modify optical, electronic and surface properties are widely lacking. To fully exploit their potential, it will be necessary to fine-tune their properties. Our main focus regarding functionalisation is studying the noncovalent interaction of the inorganic 2D materials with organic molecules and polymers. We use the optical fingerprints of both organic and nano-materials to track interactions such as doping or photoinduced charge transfer in a high throughput manner. In addition, we are also interested in covalent functionalisation at both basal planes and selectively at edge regions. While especially transition metal dichalcogenides are considered widely inert and unreactive at the basal plane, recent years have seen a significant progress and provided evidence for first proof of principle reactions that now need to be further elaborated. This will give access to a broad range of 2D materials with tailored properties which can be processed from liquids by established techniques such as spin coating, drop casting or printing. In turn, this will allow for the fabrication of multicomponent inorganic/organic hybrid systems using supramolecular chemistry.